Panobinostat (Synonyms: 帕比司他; LBH589; NVP-LBH589) 纯度: 99.20%
Panobinostat (LBH589; NVP-LBH589) 是一种有效的口服非选择性 HDAC 抑制剂,具有抗肿瘤活性。Panobinostat 可诱导 HIV-1 virus 的产生,即使在较低的浓度范围 8-31 nM,也可刺激 HIV-1 在潜伏感染细胞中的表达。Panobinostat 诱导细胞凋亡 (apoptosis) 和自噬 (autophagy)。Panobinostat 可用于难治性或复发性多发性骨髓瘤的研究。
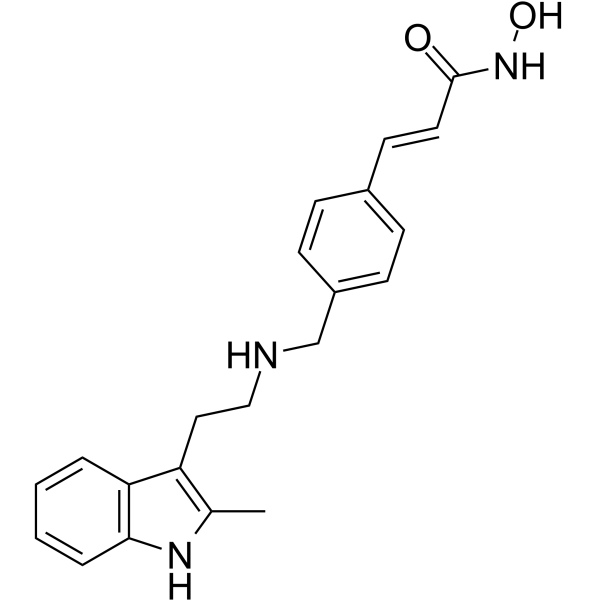
Panobinostat Chemical Structure
CAS No. : 404950-80-7
| 规格 | 价格 | 是否有货 | 数量 |
|---|---|---|---|
| Free Sample (0.1-0.5 mg) | Apply now | ||
| 10 mM * 1 mL in DMSO | ¥716 | In-stock | |
| 10 mg | ¥651 | In-stock | |
| 50 mg | ¥1850 | In-stock | |
| 100 mg | ¥3100 | In-stock | |
| 200 mg | ¥4600 | In-stock | |
| 500 mg | ¥7500 | In-stock | |
| 1 g | 询价 | ||
| 5 g | 询价 |
* Please select Quantity before adding items.
Panobinostat 相关产品
•相关化合物库:
- Covalent Screening Library Plus
- Drug Repurposing Compound Library Plus
- FDA-Approved Drug Library Plus
- FDA-Approved Drug Library Mini
- Bioactive Compound Library Plus
- Anti-Infection Compound Library
- Apoptosis Compound Library
- Cell Cycle/DNA Damage Compound Library
- Epigenetics Compound Library
- FDA-Approved Drug Library
- Histone Modification Research Compound Library
- Anti-Cancer Compound Library
- Antiviral Compound Library
- Autophagy Compound Library
- Anti-Aging Compound Library
- Drug Repurposing Compound Library
- Covalent Screening Library
- Reprogramming Compound Library
- Oxygen Sensing Compound Library
- Anti-COVID-19 Compound Library
- Orally Active Compound Library
- FDA Approved & Pharmacopeial Drug Library
- Anti-Breast Cancer Compound Library
- Anti-Lung Cancer Compound Library
- Anti-Pancreatic Cancer Compound Library
- Anti-Blood Cancer Compound Library
- Targeted Therapy Drug Library
- Anti-Liver Cancer Compound Library
- Rare Diseases Drug Library
| 生物活性 |
Panobinostat (LBH589; NVP-LBH589) is a potent and orally active non-selective HDAC inhibitor, and has antineoplastic activities[1][2]. Panobinostat induces HIV-1 virus production even at low concentration range 8-31 nM, stimulates HIV-1 expression in latently infected cells[4]. Panobinostat induces cell apoptosis and autophagy. Panobinostat can be used for the study of refractory or relapsed multiple myeloma[3]. |
||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IC50 & Target[1][5] |
|
||||||||||||||||
| 体外研究 (In Vitro) |
Panobinosta (LBH589) induces apoptosis of both MOLT-4 and Reh cells in a time- and dose-dependent manner. Panobinosta treatment results in histone (H3K9 and H4K8) hyperacetylation and regulation of cell-cycle control genes in Reh cells[1]. Panobinostat exhibites potent antiproliferative activity in human NSCLC cell lines with the IC50 ranging from 5 to 100 nM[2]. MCE has not independently confirmed the accuracy of these methods. They are for reference only. |
||||||||||||||||
| 体内研究 (In Vivo) |
Panobinosta (10, 20 mg/kg, i.p.) significantly slows tumor growth derived from Meso and NSCLC cells in vivo models. Panobinosta markedly increases acetylation of histone H3 and H4 of H69 human SCLC cells harvest from SCID mice[2]. Panobinostat (5, 10 and 20 mg/kg i.p.) demonstrates a clear benefit of decreased tumor burden, significantly improves TTE and reduces bone density loss in a disseminated multiple myeloma mouse model[3]. MCE has not independently confirmed the accuracy of these methods. They are for reference only. |
||||||||||||||||
| Clinical Trial |
|
||||||||||||||||
| 分子量 |
349.43 |
||||||||||||||||
| Formula |
C21H23N3O2 |
||||||||||||||||
| CAS 号 |
404950-80-7 |
||||||||||||||||
| 中文名称 |
帕比司他 |
||||||||||||||||
| 运输条件 |
Room temperature in continental US; may vary elsewhere. |
||||||||||||||||
| 储存方式 |
|
||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : ≥ 57 mg/mL (163.12 mM) * “≥” means soluble, but saturation unknown. 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂: ——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用; 以下溶剂前显示的百
|
||||||||||||||||
| 参考文献 |
|
| Cell Assay [1] |
Cells are washed with ice-cold PBS containing 0.1 mM sodium orthovanadate, and total proteins are isolated using RIPA lysis buffer, which includes protease inhibitors (leupeptin, antipain, and aprotinin), 0.5 mM PMSF, and 0.2 mM sodium orthovanadate. Protein amounts are quantified using the Bio-Rad protein assay. Equal amounts of proteins are loaded onto an sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel, transferred onto nitrocellulose membrane, and probed with the antibody of interest: mouse monoclonal c-Myc and mouse monoclonal p21 antibodies; rabbit polyclonal phospho-Histone H2A.X, rabbit polyclonal acetyl-Histone H3 (Lys9), and rabbit polyclonal acetyl-Histone H4 (Lys8) antibodies; mouse monoclonal p27/KIP1 antibody; mouse monoclonal anti-β-actin; and mouse monoclonal anti-GADD45G. Membranes are then washed, reprobed with appropriate horseradish peroxidase-conjugated secondary antibodies, and developed with SuperSignal chemiluminescent substrate. MCE has not independently confirmed the accuracy of these methods. They are for reference only. |
|---|---|
| Animal Administration [1] |
AE17 and TC-1 cancer cells (1×106 cells) are injected into the flanks of adult female C57Bl/6 mice and severe combined immunodeficiency (SCID) mice. M30 (10×106 cells), A549 (5×106 cells), H69 (2.5×106 cells), BK-T (6.5×106), H526 (10×106), and RG1 (10×106) cells are also injected, but in the presence of matrigel, into the flanks of SCID mice. When tumors reach 100 to 500 mm3, panobinostat is administered via i.p. injections (10-20 mg/kg) on a daily schedule (5-days-on, 2-days-off regimen) for the entire duration of the experiment. Control micereceive i.p. injections with dextrose 5% in water. Every tumor is measured with a caliper at least twice weekly. For evaluation of the effects of combination therapy on SCLC-derived tumors, SCID mice with H69 tumors are administered panobinostat. Three days after the initiation of panobinostat, and again 1 wk later, etoposide (40 mg/kg) is administered i.p. MCE has not independently confirmed the accuracy of these methods. They are for reference only. |
| 参考文献 |
|
