上海金畔生物科技有限公司为生命科学和医药研发人员提供生物活性分子抑制剂、激动剂、特异性抑制剂、化合物库、重组蛋白,专注于信号通路和疾病研究领域。
Rosiglitazone (Synonyms: 罗格列酮; BRL 49653) 纯度: 99.90%
Rosiglitazone (BRL 49653),是噻唑烷二酮类胰岛素增敏剂,也是选择性的,具有口服活性 PPARγ 激动剂,对 PPARγ1、PPARγ2 和 PPARγ 的 EC50 值分别为 30 nM、100 nM 和 60 nM。Rosiglitazone 与 PPARγ 结合,Kd 约为 40 nM。Rosiglitazone 也是 TRPC5 的激活剂 (EC50=∼30 μM) 和 TRPM3 的抑制剂。
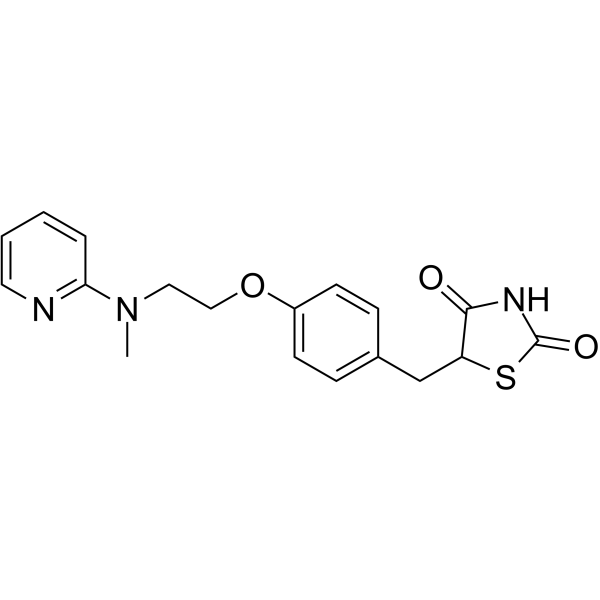
Rosiglitazone Chemical Structure
CAS No. : 122320-73-4
| 规格 | 价格 | 是否有货 | 数量 |
|---|---|---|---|
| Free Sample (0.1-0.5 mg) | Apply now | ||
| 10 mM * 1 mL in DMSO | ¥660 | In-stock | |
| 50 mg | ¥600 | In-stock | |
| 200 mg | ¥1800 | In-stock | |
| 500 mg | 询价 | ||
| 1 g | 询价 |
* Please select Quantity before adding items.
Rosiglitazone 相关产品
•相关化合物库:
- Drug Repurposing Compound Library Plus
- FDA-Approved Drug Library Plus
- FDA-Approved Drug Library Mini
- Bioactive Compound Library Plus
- Apoptosis Compound Library
- Cell Cycle/DNA Damage Compound Library
- Immunology/Inflammation Compound Library
- Membrane Transporter/Ion Channel Compound Library
- Neuronal Signaling Compound Library
- FDA-Approved Drug Library
- Anti-Cancer Compound Library
- CNS-Penetrant Compound Library
- Autophagy Compound Library
- Anti-Aging Compound Library
- Drug Repurposing Compound Library
- Diabetes Related Compound Library
- Oxygen Sensing Compound Library
- Ferroptosis Compound Library
- Anti-COVID-19 Compound Library
- NMPA-Approved Drug Library
- FDA Approved & Pharmacopeial Drug Library
- Drug-Induced Liver Injury (DILI) Compound Library
- Anti-Obesity Compound Library
- Transcription Factor Targeted Library
- Lipid Metabolism Compound Library
- Mechanoreceptors Compound Library
- Rare Diseases Drug Library
| 生物活性 |
Rosiglitazone (BRL 49653) is a selective, orally active PPARγ agonist with EC50s of 30 nM, 100 nM and 60 nM for PPARγ1, PPARγ2, and PPARγ, respectively. Rosiglitazone binds to PPARγ with a Kd of approximately 40 nM. Rosiglitazone is also an activator of TRPC5 (EC50=~30 μM) and an inhibitor of TRPM3[1][2][3][4]. |
||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IC50 & Target[1][3] |
|
||||||||||||||||
| 体外研究 (In Vitro) |
Rosiglitazone is a potent and selective activator of PPARγ, with EC50s of 30 nM and 100 nM for PPARγ1 and PPARγ2, respectively, and a Kd of appr 40 nM for PPARγ. Rosiglitazone (BRL49653, 0.1, 1,10 μM) promotes differentiation of C3H10T1/2 stem cells to adipocytes[1]. Rosiglitazone (Compound 6) activates PPARγ, with an EC50 of 60 nM[2]. Rosiglitazone (1 μM) activates PPARγ, which binds to NF-α1 promoter to activate gene transcription in neurons. Rosiglitazone (1 μM) also protects Neuro2A cells and hippocampal neurons against oxidative stress, and up-regulates BCL-2 expression in an NF-α1-dependent manner[3]. Rosiglitazone completely inhibits TRPM3 with IC50 values of 9.5 and 4.6 μM against nifedipine- and PregS-evoked activity, but such effects are not via PPARγ. Rosiglitazone inhibits TRPM2 at higher concentration, with an IC50 of appr 22.5 μM. Rosiglitazone is a strong stimulator of TRPC5 channels, with an EC50 of ~30 μM[4]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only. |
||||||||||||||||
| 体内研究 (In Vivo) |
Rosiglitazone (5 mg/kg, p.o.) decreases the serum glucose in diabetic rats. Rosiglitazone also decreases IL-6, TNF-α, and VCAM-1 levels in diabetic group. Rosiglitazone in combination with losartan increases glucose compared to diabetic and Los-treated groups. Rosiglitazone significantly ameliorates endothelial dysfunction indicated by a significantly lower contractile response to PE and Ang II and enhancement of ACh-provoked relaxation in aortas isolated from diabetic rats[5]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only. |
||||||||||||||||
| Clinical Trial |
|
||||||||||||||||
| 分子量 |
357.43 |
||||||||||||||||
| Formula |
C18H19N3O3S |
||||||||||||||||
| CAS 号 |
122320-73-4 |
||||||||||||||||
| 中文名称 |
罗格列酮 |
||||||||||||||||
| 运输条件 |
Room temperature in continental US; may vary elsewhere. |
||||||||||||||||
| 储存方式 |
|
||||||||||||||||
| 溶解性数据 |
In Vitro:
DMSO : 250 mg/mL (699.44 mM; Need ultrasonic) Ethanol : 2 mg/mL (5.60 mM; ultrasonic and warming and heat to 60°C) H2O : < 0.1 mg/mL (ultrasonic) (insoluble) 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 In Vivo:
请根据您的实验动物和给药方式选择适当的溶解方案。以下溶解方案都请先按照 In Vitro 方式配制澄清的储备液,再依次添加助溶剂: ——为保证实验结果的可靠性,澄清的储备液可以根据储存条件,适当保存;体内实验的工作液,建议您现用现配,当天使用; 以下溶剂前显示的百
|
||||||||||||||||
| 参考文献 |
|
| Kinase Assay [1] |
cDNA encoding amino acids 174-475 of PPARγ1 is amplified via polymerase chain reaction and inserted into bacterial expression vector pGEX-2T. GST-PPARγ LBD is expressed in BL21(DE3)plysS cells and extracts. For saturation binding analysis, bacterial extracts (100 μg of protein) are incubated at 4°C for 3 h in buffer containing 10 mM Tris (pH 8.0), 50 mM KCl, 10 mM dithiothreitol with [3H]-BRL49653 (specific activity, 40 Ci/mmol) in the presence or absence of unlabeled Rosiglitazone. Bound is separated from free radioactivity by elution through 1-mL Sephadex G-25 desalting columns. Bound radioactivity eluted in the column void volume and is quantitated by liquid scintillation counting[1]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only. |
|---|---|
| Cell Assay [1] |
C3H10T1/2 cells are grown in a 24-well plate in DME medium supplemented with 10% fetal calf serum. Medium and compound (Rosiglitazone) are exchanged every 3 days. Cells are stained at day 7 with Oil Red O and photographed[1]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only. |
| Animal Administration [2] |
Rats are intravenously injected with 38 mg/kg streptozotocin and after 48 h, diabetes is identified by urinary glucosuria and then random blood sugar is measured and this day is regarded as day 0. Animals with a serum glucose level of 220-300 mg/dL are selected to be used in this study. Rats are randomly separated into five groups for daily drug administration for 8 weeks: group 1: control nondiabetic rats given a vehicle only (0.5 mL/kg of 0.5% carboxy methyl celleluse orally), group 2: control diabetic rats given a vehicle, group 3: diabetic rats receiving Rosiglitazone (5 mg/kg orally), group 4: diabetic rats receiving losartan (2 mg/kg, orally), and group 5: diabetic rats receiving both Rosiglitazone and losartan[2]. 上海金畔生物科技有限公司 has not independently confirmed the accuracy of these methods. They are for reference only. |
| 参考文献 |
|
所有产品仅用作科学研究或药证申报,我们不为任何个人用途提供产品和服务